- 0 Comment
Understanding Coagulation and Flocculation in Water Treatment
Coagulation and flocculation are drinking water and wastewater treatments. They are crucial for lowering span turbidity—a turbidity measurement of water clarity—and are important for water quality as a whole. In wastewater treatment, these operations can lower suspended solids and organic
All water sources have suspended particles, such as minute colloids that are kept stable by physical forces such as static electricity. The colloids are usually negatively charged and repel each other, remaining suspended in the water for extended periods, occasionally even centuries—without settling.
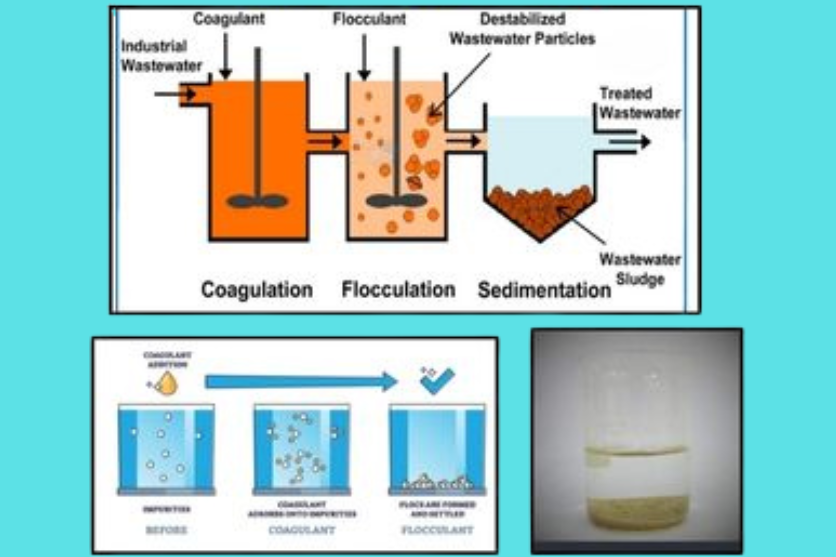
To counteract this, coagulation and flocculation are used sequentially. Coagulation neutralizes the negative charges on the particles and removes the repulsive forces. Flocculation gathers the water together gently to have these particle charges form heavy flocs, which can be removed more readily using sedimentation or filtration.
The Water Treatment Process of Coagulation—Defined
Coagulation is an essential phase of water treatment that disrupts the natural repulsive forces of small suspended particles, enabling them to aggregate and settle as large clusters. As these particles stick together, their size increases, making them simpler to remove from water. The principle of coagulants has been in practice since 2000 BC, when the Egyptians applied ground almonds coated within containers to filter
When coagulating, the particles form small clusters called micro-flocs. Although still usually invisible to the naked eye, their formation also generally leads to less cloudy surrounding water—a sign that the electrical charges holding the particles together have been neutralized. If the water remains cloudy, it could be a sign that there is a requirement for further coagulant. Overdosing, though, can turn the effect around and cause particles to repel once again from a charge polarity shift
In order to be effective, coagulation has to happen fast. Fast mixing ensures even distribution of the coagulant and encourages frequent particle collisions. Metal-based coagulant hydrolysis creates destabilizing agents in less than a second (0.01 to 1.0 seconds), so this part of the process is extremely time-sensitive. This is the reason pH correction or adding additional coagulant after dosing usually doesn’t enhance outcomes.
Although conventional back-mix reactors—vertical impellers in square tanks—are the most popular for mixing, they tend to fall. More sophisticated systems, like in-line mixers with gradient-controlled velocity, are better suited to provide optimal conditions for quick and even coagulation. These are more efficient in mixing and are more effective in the overall coagulation process.
Which Coagulant Must You Use for Water Treatment?
Metal-based coagulants, including aluminum sulfate and ferric chloride, are the most popular used in water treatment because of their ready availability, affordability, and long track record of performance. Of these, aluminum sulfate (alum) is particularly well-liked since it is cheap yet effective in precipitating suspended material and lowering turbidity.
There are other coagulants available, though, which would be more appropriate for particular applications:
Synthetic coagulants are designed chemicals with high charge density and large molecular sizes. A number of these chemicals also find applications as flocculants, depending on their composition. They are frequently applied when there is a need for improved performance or specific treatment, especially in wastewater from industries.
Natural biopolymer (renewable resource-based) coagulants come from renewable sources like plants, algae, or fungi. They are becoming popular for being less toxic, environmentally friendly, and having the potential to produce smaller amounts of sludge. While generally safer, they may have varying availability and consistency.
Finally, the selection of the coagulant relies on various considerations such as water quality, treatment objectives, environmental considerations, and cost. Proper evaluation and testing are necessary to ascertain the best and most sustainable choice for your particular water treatment requirement.
The Flocculation Water Treatment Process—Defined
Flocculation is an important stage of the water treatment process after coagulation. Once suspended solids in water are destabilized by coagulants, flocculation is a gentle but controlled agglomeration whereby these suspended particles collide and stick together to create flocs, which are larger aggregates.
This is done through the addition of flocculants, which are often polymers (cationic or anionic). The polymers consist of long chains that physically bond small particles together, making them larger and heavier. The larger the flocs, the more easily they can be separated by sedimentation, filtration, or flotation, greatly enhancing water clarity and quality
Which Flocculant Do You Need for Water Treatment?
Selecting the proper flocculant is vital in achieving efficient water and wastewater treatment. The choice relies on the suspended solids type, water chemistry, and the treatment objective. The two most widespread forms of flocculants are anionic and cationic polymers:
Cationic Flocculating Agents
Best applied to organically loaded waters, including municipal wastewater, food processing, or sludge dewatering. Act against negatively charged particles such as organic matter, bacteria, and biological solids. Assist in enhancing sludge compaction, dewatering effectiveness, and overall treatment performance. For maximum performance, most treatment systems employ a mixture of coagulants and flocculants, and performance must be verified by jar testing. Proper selection guarantees enhanced floc formation, settling is quicker, and operating expense is lower.
Anionic Flocculating Agents
Most suitable for the separation of suspended solids in industrial effluents and mining. Works effectively with positively charged particles such as mineral-based or inorganic solids. Provide rapid settling, superior water clarity, and low chemical use. Best suited for processes where water has high levels of clay, sand, or mineral solids.
Conclusion
Flocculation and coagulation are two related but differing processes that are essential to wastewater and water treatment. Coagulation counteracts the electrical charges repelling suspended particles, and flocculation aggregates them into masses that can be easily removed from the liquid phase.
At Gurupharma Chemical Industries, we provide a specifically chosen portfolio of coagulants and flocculants to ensure optimal treatment performance, reduce sludge production, and enable alternative disposal or water reuse opportunities for your operations.
Ready to optimize your water treatment process? Contact us today to discuss how our tailored solutions can assist your compliance, cost savings, and sustainability objectives.



